March 31, 2024
0 265
Unlock the Power of Continuous Learning with Udemy’s Frequent Sales
March 6, 2024
0 611
How to Ace Your Next Job Interview: Expert Insights and Strategies
March 6, 2024
0 344
Mastering the Trio: Project, Program & Portfolio Management
February 23, 2024
0 1,312
The Evolution of Performance Reviews: From Annual Appraisals to Continuous Feedback
February 14, 2024
0 454
Behaviors that kill employee motivation
February 8, 2024
0 3,577
Free Udemy Courses 100% Coupon
December 17, 2021
1 7,504
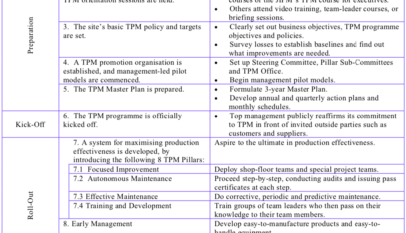
Getting Ready for TPM
September 15, 2021
0 2,911
Chapter 12. Completing and Upgrading the TPM Programme (Levels 1, 2 and 3). Part 1
5S
Autonomous Maintenance
Blog
Change Management
Early Equipment Management
Early Product Management
Education and Training
Focused Improvement
Instructor Training
Manufacturing Support
Planned Maintenance
Quality
Safety, Health and Environment
TPM
TPM Introduction
World Class Manufacturing
April 30, 2021
0 16,646
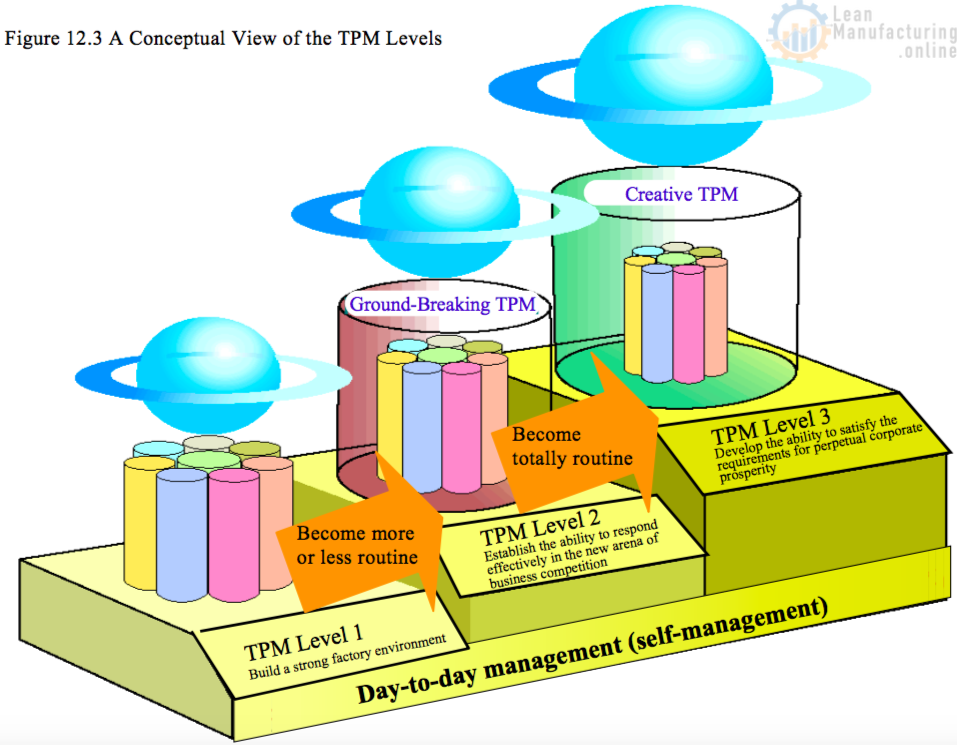
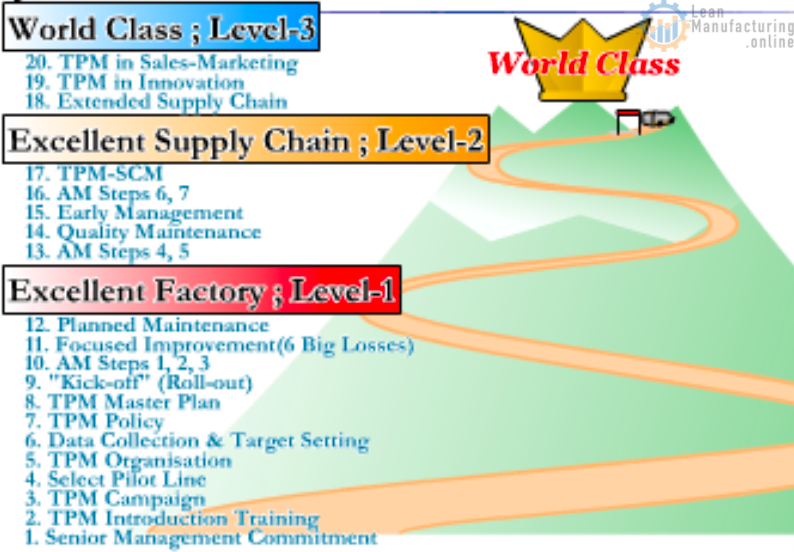
TPM 3 Levels – path to World-Class Manufacturing
December 1, 2020
0 2,227
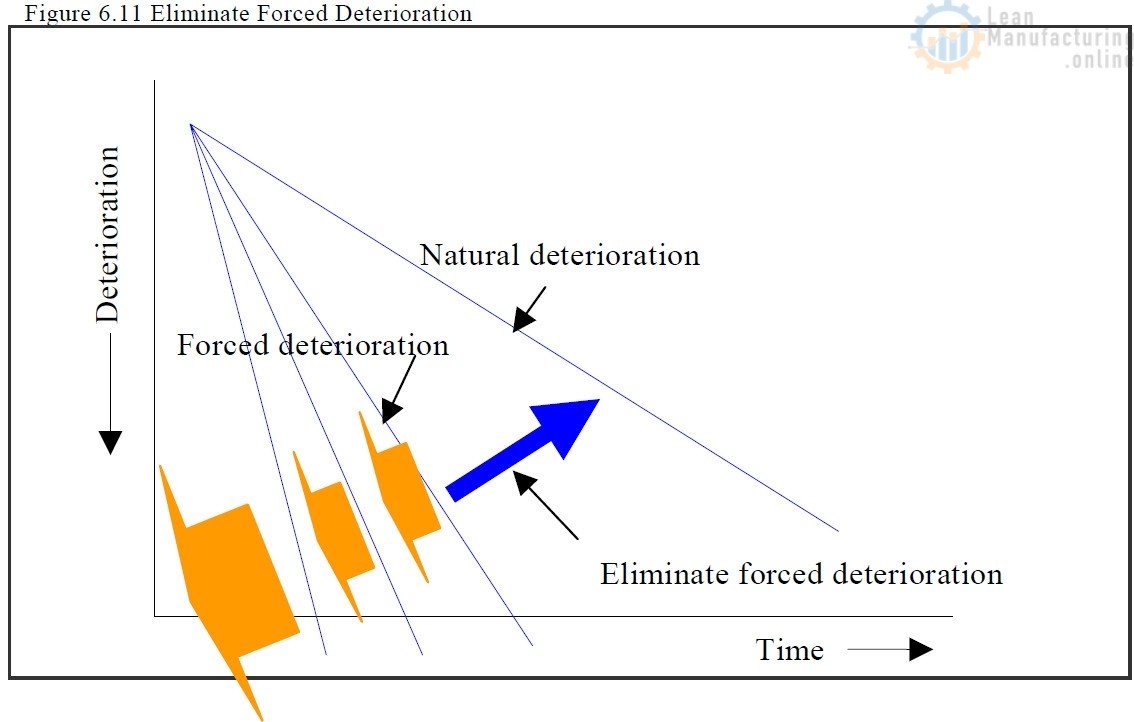
Chapter 6. Planned (Effective) Maintenance. Part 3
December 1, 2020
0 2,059
Chapter 6. Planned (Effective) Maintenance. Part 2
December 1, 2020
0 5,836
Chapter 6. Planned (Effective) Maintenance. Part 1
Most Popular
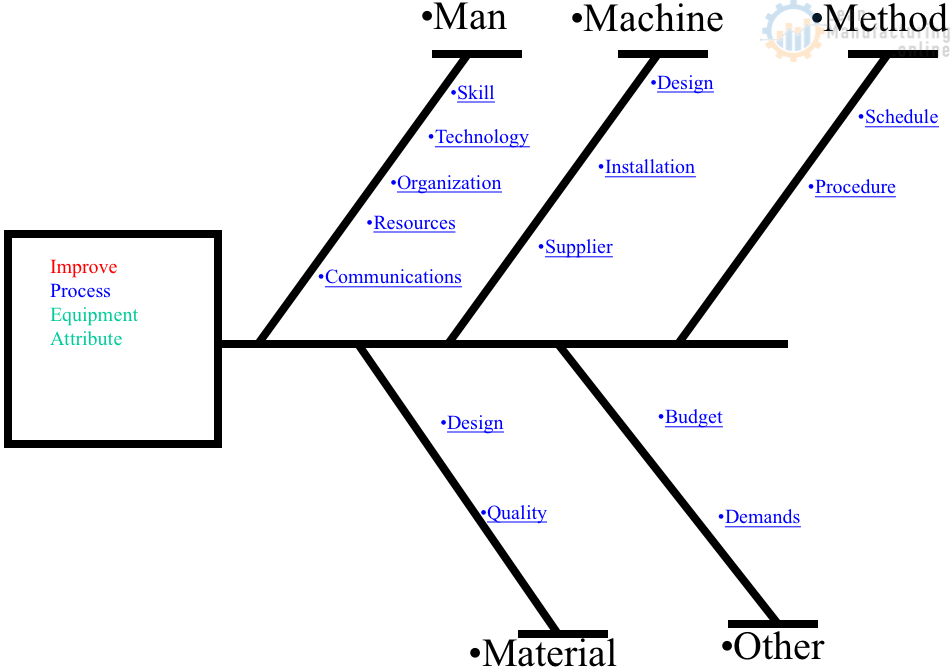
4M Analysis Process
The purpose of this procedure is to define the steps to do a 4M …