Early Equipment Management
Unlocking the Secrets of Pharmaceutical Validation: A Comprehensive Training Guide
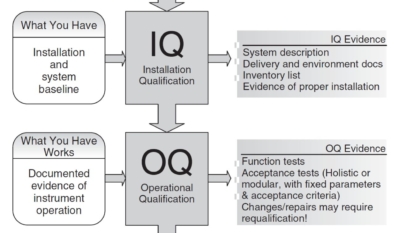
Discover the critical stages of pharmaceutical validation: Design Qualification (DQ), Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ). Dive into our comprehensive guide that serves as a full training session on ensuring compliance and performance in the life sciences field.
Read More »TPM Pillar Self Assessment
5S Autonomous Maintenance Early Equipment Management Education and Training Focused Improvement Manufacturing Support Planned Maintenance Quality Safety, Health and Environment TPM World Class Manufacturing
The TPM Self Assessment’s purpose is to provide information to assist in running the factory. By showing the strengths and weaknesses of each TPM pillar using a structured approach.
Read More »Example of Food Manufacturing site review
5S Autonomous Maintenance Early Equipment Management Education and Training Manufacturing Support Planned Maintenance Quality Safety, Health and Environment World Class Manufacturing
Master Plan Show the results for the main objectives with consistent benchmarks and targets – Breakdowns, Minor Stops, Quality Defects and ZERO loss time accidents.Highlight why the Site needs TPM. “TPM is not the objective; it is the way to deliver the objectives.”Each pillar must have key indicators to link…
Read More »Steps 4 to 7: fabrication, manufacturer’s test-run, installation, and commissioning
EEM commissioning control step.
Read More »