Quality
Mastering Acceptance Quality Level (AQL): A Practical Guide
Acceptance Quality Level (AQL) is a widely used quality control tool that helps manufacturers and suppliers strike the right balance between product quality and inspection efficiency. This guide explains what AQL is, how it works, its sampling process, inspection levels, and when to use it effectively.
Read More »The Complete Guide to GD&T: Symbols, Rules, and Best Practices
Master GD&T and eliminate ambiguity in design and manufacturing. Learn symbols, MMC, LMC, tolerance zones, and practical tips for precision engineering.
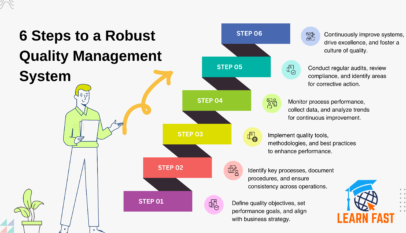
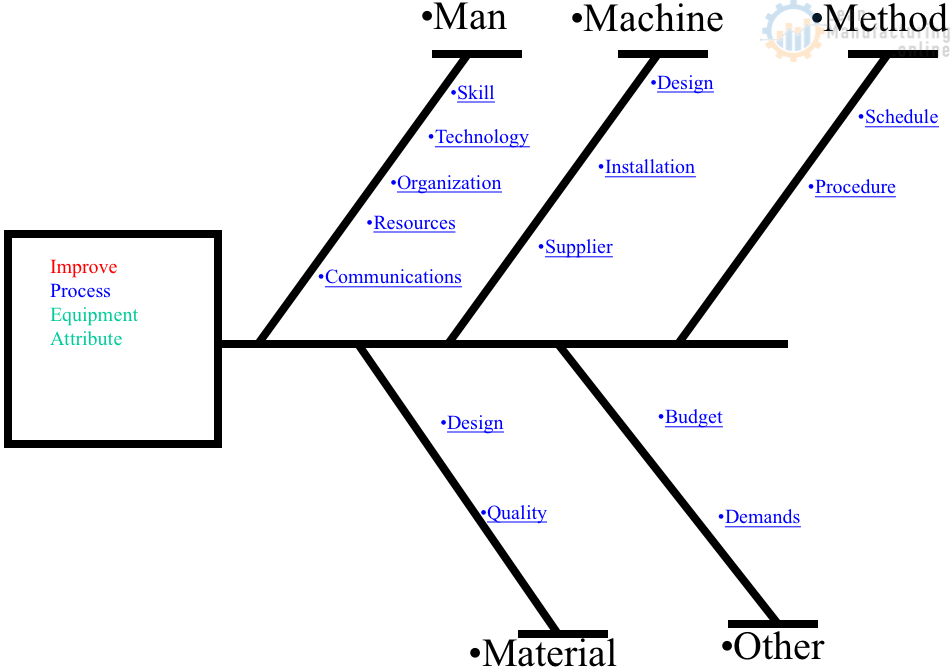
Read More »Essential Quality Tools for Effective Process Improvement
Discover essential quality tools to improve your process efficiency and ensure product reliability. Learn about key methodologies, their applications, and benefits.
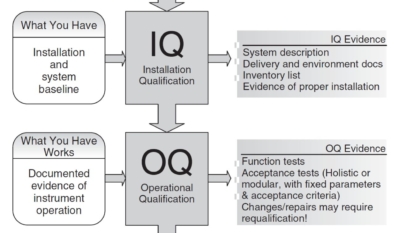
Read More »Unlocking the Secrets of Pharmaceutical Validation: A Comprehensive Training Guide
Discover the critical stages of pharmaceutical validation: Design Qualification (DQ), Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ). Dive into our comprehensive guide that serves as a full training session on ensuring compliance and performance in the life sciences field.
Read More »